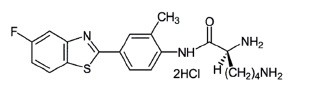
Antitumoral Phortress Small Molecule (Tool Compound)
The Antitumoral Phortress compound is a potential anticancer agent for treatment of human breast carcinoma. Phortress is highly selective for susceptible cancer cells because of its mechanism of action.