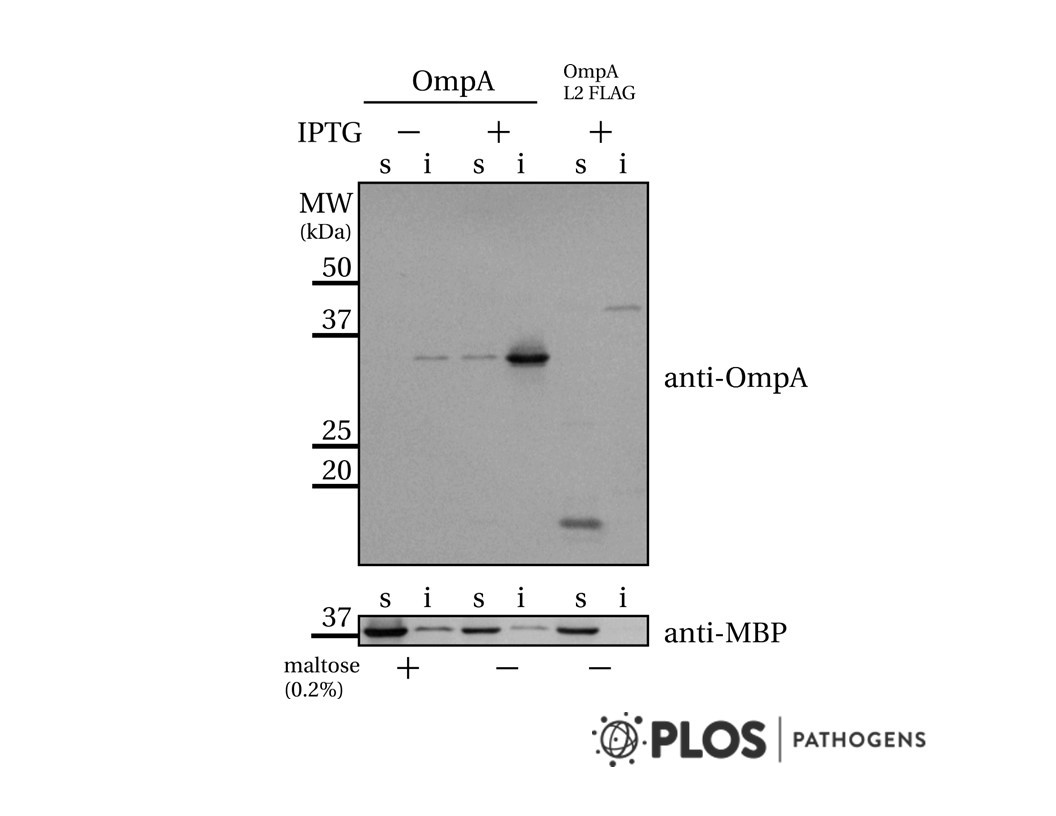
Anti-MBP [R29.6]
R29.6 is useful for detection and isolation of recombinant MBP fusion proteins.
Contributors
| Inventor | Institute |
|---|---|
| Julian Gannon | Cancer Research UK, London Research Institute: Clare Hall Laboratories |
| Cat. #: | 151017 |
|---|---|
| Tool sub type: | Primary antibody |
| Unit size: | 100 ug |
| Research Fields: | Cell biology;Neurobiology |
| Application: | ChIP ; IHC ; IF ; IP ; WB |
| Target: | Maltose binding protein (MBP) |
| Reactivity: | Bovine |
| Clone: | R29.6 |
| Host: | Mouse |
| Class: | Monoclonal |
| Alternate name: | CCNA1; Cyclin A1; Testicular Tissue Protein Li 34; CT146 |
|---|---|
| Product description: | R29.6 is useful for detection and isolation of recombinant MBP fusion proteins. |
| Conjugation: | Unconjugated |
| Isotype: | IgG1 |
| Molecular weight: | 53 kDa |
| Immunogen: | MOS maltose binding protein fusion protein |
| Myeloma used: | Sp2/0-Ag14 |
| Target background: | MBP is a bacterial protein commonly used as a fusion protein. |
|---|
| Format: | Liquid |
|---|---|
| Concentration: | 1 mg/ml |
| Storage buffer: | PBS with 0.02% azide |
| Storage conditions: | '-15° C to -25° C |
| Shipping conditions: | Dry ice |
| References: |
Verhoeven et al. 2009. PLoS One. 4(8):e6739. PMID: 19707582. Differential bacterial surface display of peptides by the transmembrane domain of OmpA. Im et al. 2009. Dev Cell. 17(2):234-43. PMID: 19686684. Structure and function of the ESCRT-II-III interface in multivesicular body biogenesis. Liu et al. 2006. Genome Res. 16(12):1517-28. PMID: 17053089. Whole-genome comparison of Leu3 binding in vitro and in vivo reveals the importance of nucleosome occupancy in target site selection. Liu et al. 2005. Genome Res. 15(3):421-7. PMID: 15710749. DIP-chip: rapid and accurate determination of DNA-binding specificity. |
|---|
Images
View GalleryRelated Tools for Antibodies
| Cat. # | Tool Name | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 151021 | Anti-CyclinB1 [V152] |
Key Info
Anti-CyclinB1 [V152]
|
View Tool | |||||||||||||||||||
| 151023 | Anti-IL12 [1-1D5] |
Key Info
Anti-IL12 [1-1D5]
|
View Tool | |||||||||||||||||||
| 151036 | Anti-ICAM1 [15.2] |
Key Info
Anti-ICAM1 [15.2]
|
View Tool | |||||||||||||||||||
| 151039 | Anti-Integrin aVb3 [23C6] |
Key Info
Anti-Integrin aVb3 [23C6]
|
View Tool | |||||||||||||||||||
| 151018 | Anti-Mos [R38.1] |
Key Info
Anti-Mos [R38.1]
|
View Tool | |||||||||||||||||||