US/VOT-N33 Cell Line
Migrating neuroblasts, bipolar neuronal phenotypes
Contributors
| Inventor | Institute |
|---|---|
| Matthew C Holley | University of Sheffield |
| Cat. #: | 153626 |
|---|---|
| Tool sub type: | Continuous |
| Unit size: | 1×10^6 cells / vial |
| Research Fields: | Developmental biology;Drug development;Genetics |
| Organism: | Mouse |
| Tissue: | Embryonic |
| Model: | Transgenic |
| Morphology: | Neuronal |
| Growth properties: | Adherent |
| Primary citation: | Lawoko-Kerali et al. 2004. Dev Dyn. 231(4):801-14. PMID: 15499550 |
| Alternate name: | US/VOT-N33; Ventral Otocyst-Neuroblast cell line number 33; University of Sheffield/Ventral OTocyst-Neuroblast 33; VOT-N33 |
|---|---|
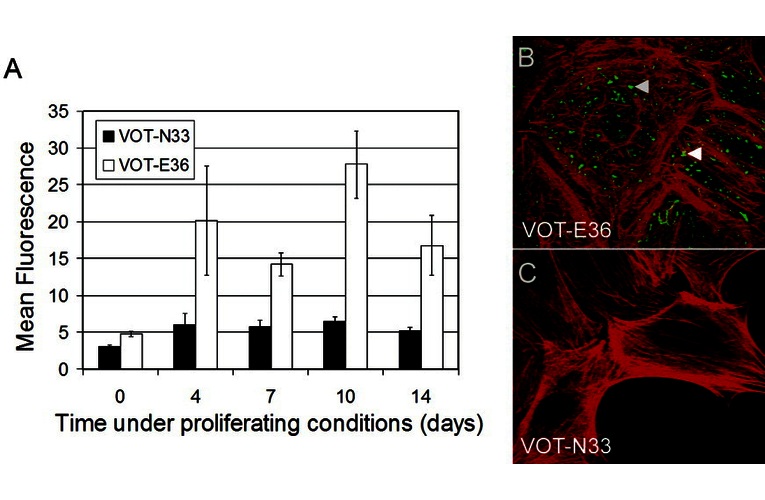
| Product description: | US/VOT-N33 Cell Line is a conditionally immortalized cell line representing migrating neuroblasts derived from the ventral otocyst of the Immortomouse™ at embryonic day 10.5 (E10.5). At this developmental stage, the sensory epithelia of the inner ear have not yet differentiated, and the ventral otocyst epithelium is competent to give rise to auditory neurons and epithelial cells. The cell line was established to model early inner ear development and mechanisms underlying hearing loss in mammals. The cells carry a stable insertion of the temperature-sensitive SV40 large T antigen (H-2Kb-tsA58) under the control of a γ-interferon-sensitive MHC Class I promoter, allowing conditional immortalization. The growth temperature and presence or absence of γ-interferon are critical for phenotype: Proliferating conditions: 33°C with γ-interferon Differentiating conditions: 39°C without γ-interferon The cell line has been extensively characterized using Affymetrix mouse microarrays and has demonstrated functional integration following transplantation to the cochlear nerve in vivo. |
| CRISPR: | No |
| Conditional: | Yes |
| Conditional description: | The cells were derived from ImmortomiceTM (Jat et al 1991 Proc. Nat. Acad. Sci. USA 88, 5096-5100) carrying a stable insertion of the conditional immortalising gene H-2Kb-tsA58, which describes a temperature-sensitive variant of the SV40 immortalising gene that encodes the large tumour antigen under the control of the γ–interferon-sensitive MHC Class 1 promoter. The cells proliferating culturing conditions: 33°C with γIFN; and the cells differentiating culturing conditions: 39°C without γIFN |
| Production details: | Homozygous male Immortomice™ (originally derived from injected oocytes of CBA/Ca × C57BL/10 mice) were time-mated with wild-type C57BL/6 female mice to produce heterozygous offspring. Embryos were harvested at E10.5, and ventral otocysts were dissected under sterile conditions. Explants were cultured in medium containing 50 U/mL γ-interferon at 33°C to support expression of the conditional immortalizing gene (H-2Kb-tsA58). Selection of explants was based on expression of tissue-specific markers, including the transcription factor GATA3, under differentiating conditions in vitro |
| Cellosaurus ID: | CVCL_VC37 |
| Model description: | Conditionally immortalized cell line |
| Disease: | Deafness |
| Format: | Frozen |
|---|---|
| Storage conditions: | Liquid Nitrogen |
| Shipping conditions: | Dry ice |
| Growth medium: | MEM with 10% FCS, 50Units/ml y-IFN, L-glutamine, 33°C |
| Temperature: | 33°C |
| Mycoplasma free: | Yes |
| Biosafety level: | 1 |
| References: |
Sekiya et al. 2007. Eur J Neurosci. 25(8):2307-18. PMID: 17445229 Lawoko-Kerali et al. 2004. Dev Dyn. 231(4):801-14. PMID: 15499550 |
|---|
Images
View GalleryRelated Tools for Cell lines
| Cat. # | Tool Name | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 151425 | TR146 Cell Line |
Key Info
TR146 Cell Line
|
View Tool | |||||||||||||
| 151445 | 1-7HB2 Cell Line |
Key Info
1-7HB2 Cell Line
|
View Tool | |||||||||||||
| 151558 | MEF Trex1 KO Cell Line |
Key Info
MEF Trex1 KO Cell Line
|
View Tool | |||||||||||||
| 151579 | U-2 OS Gal4-p300 Cell Line |
Key Info
U-2 OS Gal4-p300 Cell Line
|
View Tool | |||||||||||||
| 151629 | MEF Raf1 FF/FF KI Cell Line |
Key Info
MEF Raf1 FF/FF KI Cell Line
|
View Tool | |||||||||||||