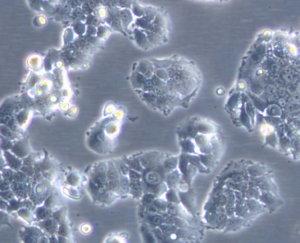
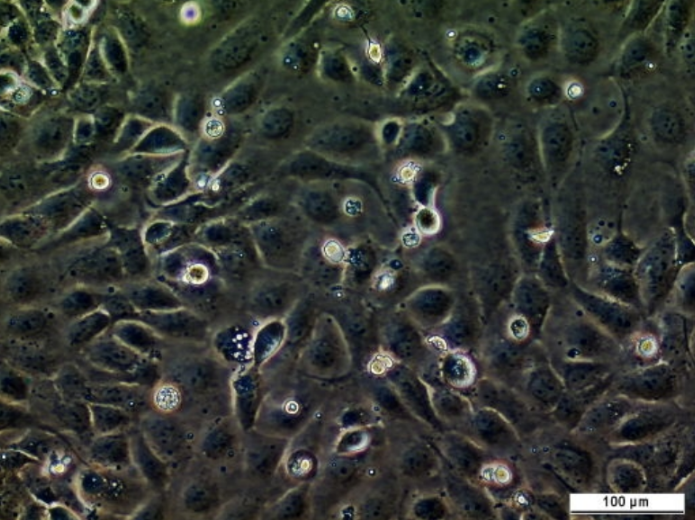
MCF7/182R-6 Cell Line
The MCF7/182R-6 Cell line is a breast cancer cell line resistant to fulvestrant. Treatment with the steroidal antiestrogen fulvestrant has proven effective upon progression on tamoxifen therapy and is now approved for second-line treatment after tamoxifen or aromatase inhibitors. As for tamoxifen treatment of advanced breast cancer, resistance will inevitably occur also for fulvestrant.
Contributors
| Inventor | Institute |
|---|---|
| Anne Lykkesfeldt | Danish Cancer Society, Denmark |
| Cat. #: | 152105 |
|---|---|
| Tool sub type: | Continuous |
| Unit size: | 1×10^6 cells / vial |
| Cancer types: | Breast cancer |
| Research Fields: | Cancer;Drug development |
| Organism: | Human |
| Tissue: | Breast |
| Gender: | Female |
| Model: | Cancer cell line |
| Growth properties: | Adherent |
| Cancer Types In Detail: | Breast cancer;Fulvestrant resistant |
| Primary citation: | Lykkesfeldt et al. 1995. Int J Cancer. 61(4):529-534. PMID: 7759159. |
| Alternate name: | MCF-7/182R-6 |
|---|---|
| Product description: | The MCF7/182R-6 cell line is a breast cancer cell line resistant to fulvestrant (Faslodex). The MCF7/182R-6 cell line is a human breast cancer cell line established from a clone of MCF7/S0.5 cells surviving long term growth with the pure steroidal antiestrogen ICI 182,780 (fulvestrant) in 100 nM concentration. The cellular classification is epithelial, and their shape is polygonal. MCF7/182R-6 cells express oestrogen receptor alpha and do not express progesterone receptor. Treatment with the steroidal antioestrogen fulvestrant has proven effective upon progression on tamoxifen therapy and is now approved for second-line treatment after tamoxifen or aromatase inhibitors. As for tamoxifen treatment of advanced breast cancer, resistance will inevitably occur also for fulvestrant. Clarification of the molecular changes associated with the resistant growth is needed to find targeted treatments to resistant tumour cells and treatments that can inhibit or delay the emergence of resistance. |
| Gender: | Female |
| Conditional: | Yes |
| Production details: | The MCF7/182R-6 cell line has been established from a clone of MCF7/S0.5 cells surviving long term growth with the pure steroidal antiestrogen ICI 182,780 in 100 nM concentration, see Lykkesfeldt et al (1995). The MCF7/182R-6 cells are grown in the presence of fulvestrant. |
| Cellosaurus ID: | CVCL_W536 |
| Parental cell line: | MCF7 S0.5 |
| Disease: | Cancer |
| Format: | Frozen |
|---|---|
| Storage conditions: | Liquid Nitrogen |
| Shipping conditions: | Dry ice |
| Growth medium: | Phenol red free DMEM/F12 (1:1) supplemented with 1% FCS, Glutamax 2,5 mM and 6 ng/ml insulin. Supplemented with 100nM fulvestrant to maintain resistance. |
| Subculture routine: | After thawing, dilute the cell suspension with sufficient medium and distribute 5 mL each into T25 flasks to achieve a seeding density of 0.5-1.0 x 10^4 / cm2. Place in a 37°C, 5% CO₂ incubator. Change medium after 24 hours to remove residual DMSO and then every 2-3 days. Subculture routine: Split 1:30 weekly with Trypsin-EDTA for detachment at 37 °C for 5 minutes. Please also see detailed protocol within the Product Datasheet in the Documentation section below. |
| Temperature: | 37° C |
| Atmosphere: | 5% CO2 |
| Storage medium: | 10% DMSO in FCS. |
| Mycoplasma free: | Yes |
| Biosafety level: | 1 |
| References: |
Thrane et al. 2015. Oncogene. 34:4199–4210. PMID: 25362855. Sonne-Hansen et al. 2010. Breast Cancer Res Treat. 121(3):601-613. PMID: 19697122. Frogne et al. 2008. Breast Cancer Res Treat. 114(2):263-275. PMID: 18409071. Frankel et al. 2006. Breast Cancer Res Treat. 104(2):165-179. PMID: 17061041. Frogne et al. 2005. Endocr Relat Cancer. 12(3):599-614. PMID: 16172194. Nabha et al. 2005. Oncogene. 24(19):3166-3176. PMID: 15735693. Lykkesfeldt et al. 1995. Int J Cancer. 61(4):529-534. PMID: 7759159. |
|---|
Images
View GalleryRelated Tools for Cell lines
| Cat. # | Tool Name | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 151014 | MCF7 AREc32 Cell Line |
Key Info
MCF7 AREc32 Cell Line
|
View Tool | |||||||||||||
| 151446 | FLYRD18 Cell Line |
Key Info
FLYRD18 Cell Line
|
View Tool | |||||||||||||
| 151447 | CMT 64 Cell Line |
Key Info
CMT 64 Cell Line
|
View Tool | |||||||||||||
| 151479 | HCT 116 p300 KO [D10] Cell Line |
Key Info
HCT 116 p300 KO [D10] Cell Line
|
View Tool | |||||||||||||
| 151630 | PDVC57B Cell Line |
Key Info
PDVC57B Cell Line
|
View Tool | |||||||||||||